Broomrapes (Orobanche and Phelipanche spp.) are obligate plant parasites with a broad range of agricultural crop hosts. In non-parasitic plant species, seeds generally initiate germination when exposed to favorable conditions of temperature, humidity, oxygen, and, occasionally, light. However, for obligate parasitic plants like broomrapes, a chemical signal from the host plant is essential. Germination of broomrape can only occur under appropriate soil conditions and when the seed receives a strigolactone chemical signal released from the roots of a suitable host. Strigolactones (SLs) are carotenoid-derived hormones that play a crucial role in various aspects of plant growth and development. Fertilizers can regulate the production of these plant hormones (Xie et al. 2010).
Fertilization can improve soil conditions and lead to reduced initiation of broomrape parasitism (Fernández-Aparicio et al. 2016). Studies have shown that heavy infestations of crenate broomrape (Orobanche crenata Forsk.) on faba beans are linked to lower soil fertility (Trabelsi et al. 2017), and parasitism of Egyptian broomrape on tomato occurs more frequently in low-nutrient conditions (Jain and Foy 1992). The application of fertilizers has been reported to suppress the occurrence of other parasitic plants such as Striga (Jamil et al. 2011) and Egyptian broomrape (Phelipanche aegyptiaca) (Jain and Foy 1992). Fertilizers can reduce parasitism and enhance crop tolerance both directly, through toxic effects, and indirectly by improving soil fertility and plant health.
Direct toxic effect of fertilizers
Nutrient management can enhance both resistance and tolerance to broomrape parasitism in crops at the pre-attachment and post-establishment stages. Increasing the levels of nitrogen (N) and phosphorus (P) in the soil through fertilizer application can reduce the germination and subsequent infestation rates of parasitic weeds (Jamil et al. 2011). Ammonium nitrate combined with potassium phosphate or the use of ammonium phosphate alone proved to be effective in reducing parasitism and promoting the growth of tomato plants compared to potassium sulfate (Jain and Foy 1992). The direct inhibitory effects of nutrients on broomrape seeds can occur during the preconditioning, germination, and seedling elongation stages. Preconditioning Egyptian broomrape seeds in the presence ofammonium salts, such as ammonium sulfate or urea, significantly inhibited their germination; in contrast, nitrate did not have the same inhibitory effect (Jain and Foy 1992). Increasing nitrogen rate (ammonium nitrate) decreased seed germination and radicle length of branched broomrape (Irmaileh 1994). Another experiment showed that nitrogen in the ammonium form resulted in greater inhibition than nitrate, and the inhibition mechanism was actually a reduction in radicle elongation rather than inhibition of germination (Westwood and Foy. 1999).
Down-regulating of Strigolactones (SLs)
Fertilization can protect crops from parasitism by downregulating the synthesis and exudation of strigolactones, which are the most potent germination-inducing factors for root parasites (Fernández-Aparicio et al. 2016). Plants release SLs in different situations, including the establishment of symbiotic relationships between plants and certain soil microorganisms (Besserer et al. 2006; Kapulnik and Koltai 2014) and during stress response (Kapulnik and Koltai 2014). It is likely that plants produce strigolactones as a "cry for help," which broomrape exploits to its advantage. The availability of nutrients, particularly nitrogen, can decrease plant stress and subsequently downregulate the production of strigolactones. Effects of N, P, and K deficiencies on SL production showed that both N and P deficiencies enhanced SL exudation in resistant genotypes of faba bean (Trabelsi et al. 2017) and red clover (Yoneyama et al. 2012), while K deficiency had no effect (Trabelsi et al. 2017). A similar positive effect of low phosphate levels on SL production was also observed in tomato (López-Ráez et al. 2008).
In summary, effective nutrient management is a vital strategy in reducing broomrape parasitism and enhancing crop tolerance. By manipulating soil fertility and nutrient availability, it is possible to directly inhibit broomrape development and indirectly protect crops by downregulating strigolactone production. Appropriate fertilization and other stress-reducing management practices can reduce broomrape parasitism.
In addition to the indirect effects on broomrape, researchers in the Hanson lab are investigating the direct toxic effects of various fertilizers on broomrape seeds during three stages: preconditioning, germination, and post-germination (Figure 1). In future studies, we aim to explore the indirect effects of fertilization on broomrape parasitism, focusing on how nutrient management can influence the production of strigolactones and other related mechanisms. The ultimate goal of this work is to determine if manipulating fertilizer form, timing, or rates could directly inhibit branched broomrape and maximize tomato resilience to broomrape parasitism as part of an integrated management strategy.
 |
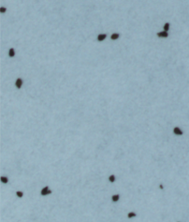 |
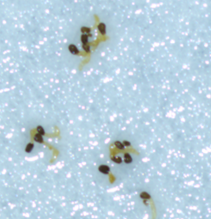 |
| a | b | c |
Figure 1: Branched broomrape seeds in different treatment conditions:
a) Germination (elongated radicle) observed in the control group.
b) No germination was observed when ammonium phosphate was applied at the germination stage.
c) Elongated radicle changed color when ammonium phosphate was applied post-germination.
References
Besserer, A., Puech-Pagès, V., Kiefer, P., Gomez-Roldan, V., Jauneau, A., Roy, S., ... & Séjalon-Delmas, N. (2006). Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biology, 4(7), e226.
Fernández-Aparicio, M., Reboud, X., & Gibot-Leclerc, S. (2016). Broomrape weeds. Underground mechanisms of parasitism and associated strategies for their control: a review. Frontiers in Plant Science, 7, 171714.
Irmaileh, B. A. (1994). Nitrogen reduces branched broomrape (Orobanche ramosa) seed germination. Weed Science, 42(1), 57-60.
Jain, R., & Foy, C. L. (1992). Nutrient effects on parasitism and germination of Egyptian broomrape (Orobanche aegyptiaca). Weed Technology, 6(2), 269-275.
Jamil, M., Charnikhova, T., Cardoso, C., Jamil, T., Ueno, K., Verstappen, F., ... & Bouwmeester, H. J. (2011). Quantification of the relationship between strigolactones and Striga hermonthica infection in rice under varying levels of nitrogen and phosphorus. Weed Research, 51(4), 373-385.
Kapulnik, Y., & Koltai, H. (2014). Strigolactone involvement in root development, response to abiotic stress, and interactions with the biotic soil environment. Plant Physiology, 166(2), 560-569.
Trabelsi, I., Yoneyama, K., Abbes, Z., Amri, M., Xie, X., Kisugi, T., ... & Kharrat, M. (2017). Characterization of strigolactones produced by Orobanche foetida and Orobanche crenata resistant faba bean (Vicia faba L.) genotypes and effects of phosphorous, nitrogen, and potassium deficiencies on strigolactone production. South African Journal of Botany, 108, 15-22.
Westwood, J. H., & Foy, C. L. (1999). Influence of nitrogen on germination and early development of broomrape (Orobanche spp.). Weed Science, 47(1), 2-7.
Xie, X., Yoneyama, K., & Yoneyama, K. (2010). The strigolactone story. Annual Review of Phytopathology, 48, 93-117.
Yoneyama, K., Xie, X., Kim, H. I., Kisugi, T., Nomura, T., Sekimoto, H., ... & Yoneyama, K. (2012). How do nitrogen and phosphorus deficiencies affect strigolactone production and exudation?. Planta, 235, 1197-1207.